Description
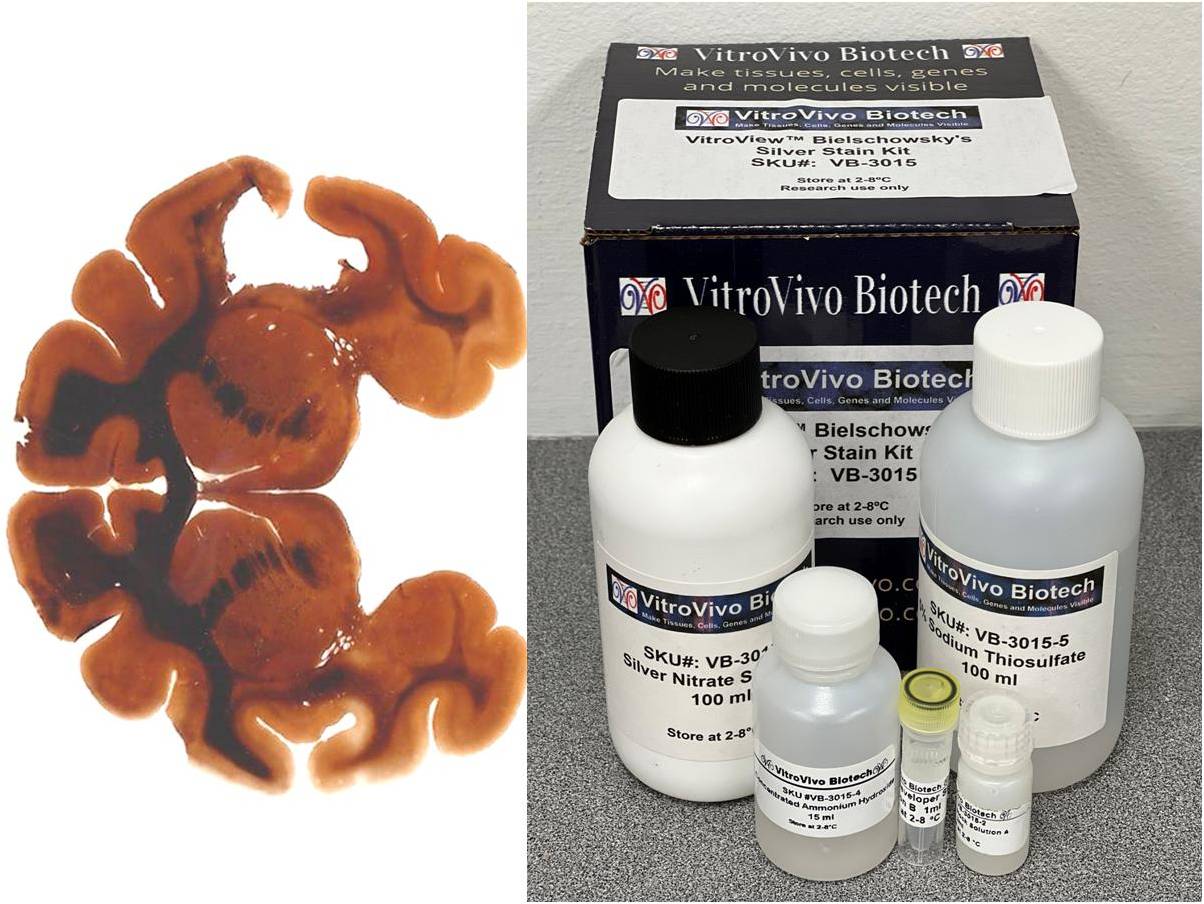
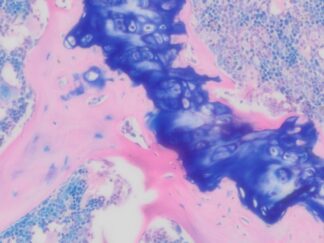
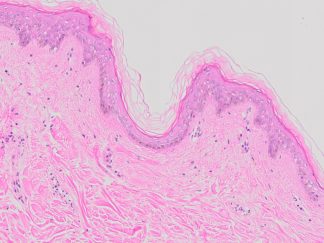
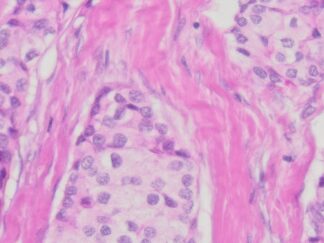
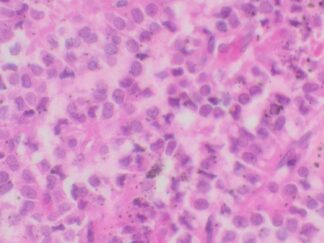
Achieve exceptional clarity in neurohistology with the Bielschowsky’s Silver Stain Kit. Designed for the precise staining of neuronal fibers and axons, this kit simplifies the complex silver impregnation process to deliver consistent, high-contrast visualization of critical pathological markers like senile plaques and neurofibrillary tangles. Whether for routine diagnostics or advanced neurodegenerative research, our kit provides the reliability and structural resolution your lab demands.
Kit Contents
| VB-3015-1 | Silver Nitrate Solution (20%) | 100 ml |
| VB-3015-2 | Developer Stock Solution | 5 ml |
| VB-3015-3 | Concentrated Ammonium Hydroxide | 20 ml |
| VB-3015-4 | 5% Sodium Thiosulfate Solution | 100 ml |
Storage Condition:
Store at 2-8ºC.
Protocol
- Let the kit stand at room temperature for 30 min before use.
- Section Preparation:
1) Paraffin-embedded tissue sections: Incubate sections in xylene for 6 minutes, repeating this step twice. Subsequently, immerse in 100% ethanol for 2 minutes, followed by two immersions in 95% ethanol for 2 minutes each. Proceed with immersion in 70% ethanol for 2 minutes. Rinse sections thoroughly in deionized water (dH2O) for 5 minutes.
2) Frozen sections: Allow sections to air dry for 5–10 minutes, then rinse in dH2O for 5 minutes
- Prepare working Ammoniacal Silver Solution immediately before use:
1) Place 20 ml of 20% silver nitrate in a glass beaker.
2) Add concentrated ammonium hydroxide dropwise. A brown precipitate will form first. Continue adding ammonia until the precipitate just dissolves, leaving a clear solution.
3) (Option) Add 5-10 drops of 20% silver nitrate to re-establish slight opalescence.
4) (Option) Filter before use.Critical note: Do not over-add ammonia; overstabilization reduces staining.
- Incubate slides in the ammonium silver solution in a 37-40 ºC oven or water bath for 20-30 minutes in dark.
- Prepare developer working solution by mixing the 1 ml of Developer Stock Solution in 100ml dH2O. This solution should be prepared fresh for each staining experiment and discarded after use.
- Ethanol Rinse: Perform 2 quick dips of slides from step 4 in 95% Ethanol (approx. 10 seconds total) to remove excess silver. Note: Do not use water, as it may prematurely wash out the stain or cause background artifacts.
- Place slides directly (do not wash slides) in developer working solution for -1-5 minute. The staining reaction can occur quickly, so we recommend first determining the appropriate incubation time using a test slide and visualizing it under a microscope.
- Dip slides for 0.5-1 minute in 1% ammonium hydroxide solution (1ml concentrated ammonium hydroxide in 100ml dH2O) to stop the silver reaction.
- Wash slides 3 × with dH2O.
- Place slides in 5% sodium thiosulfate solution for 5 minutes.
- Wash slides in running tap water for 5 minutes.
- Dehydrate slides 2 × 2 minutes in 95% ethanol.
- Dehydrate slides 2 × 2 minutes in 100% ethanol.
- Incubate slides 2× 5 minutes in xylene.
- Mount coverslip onto glass slide with Permount or other suitable organic mounting medium.
Expected results
- Axons, neurofibrillary tangles and senile plaques ——- black
- Background —————————————————yellow to brown
Positive Controls : Brain tissue
More Images
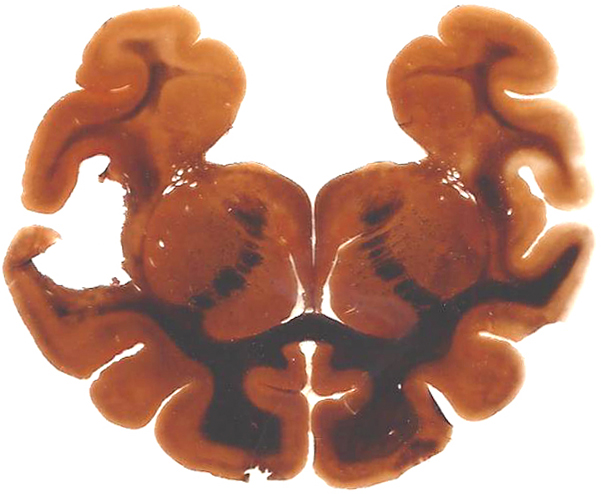
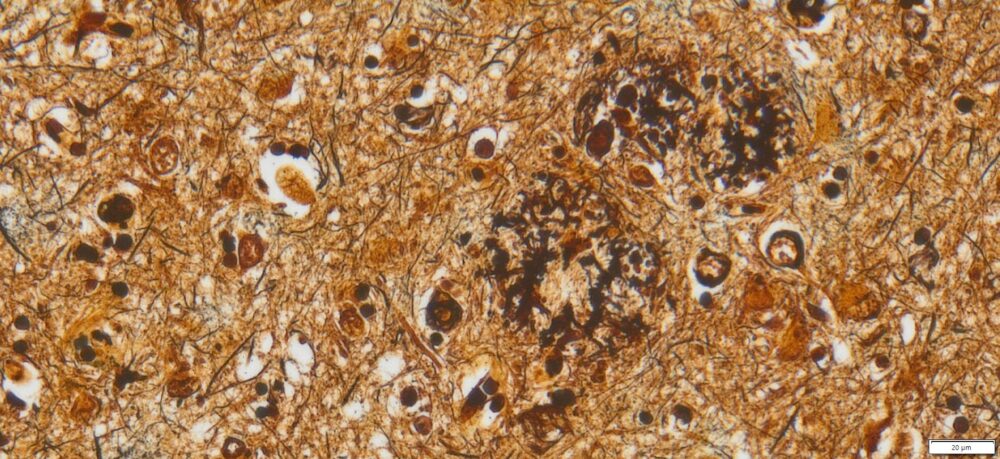
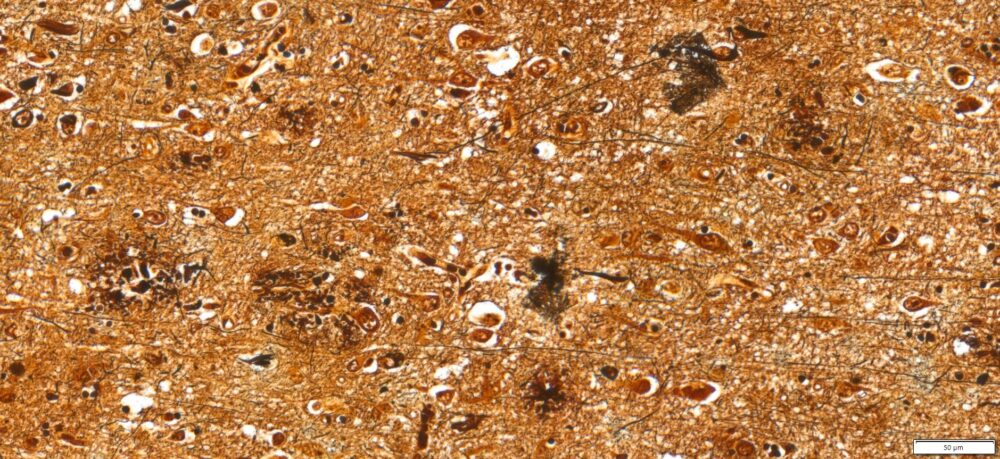
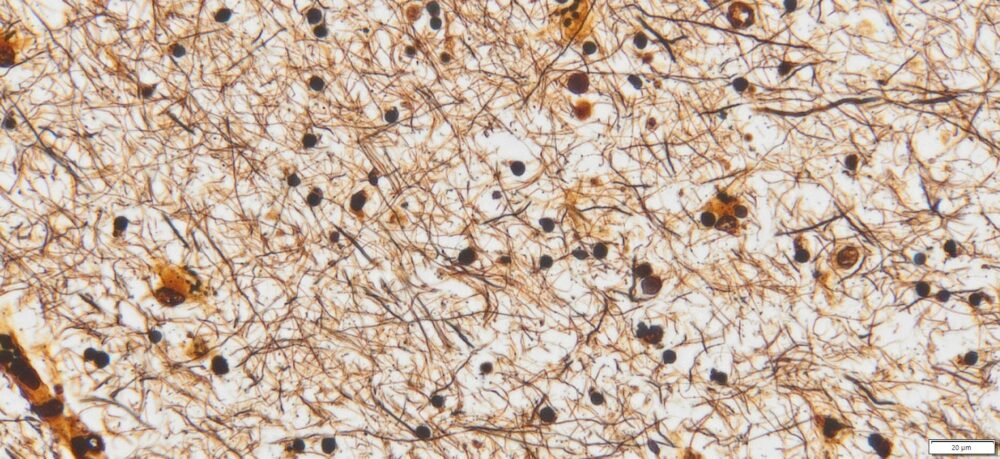
Note: This Bielschowsky’s Silver Stain Kit is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.
Precautions: Handle with care. Avoid contact with eyes, skin and clothing. Do not ingest. Wear gloves.