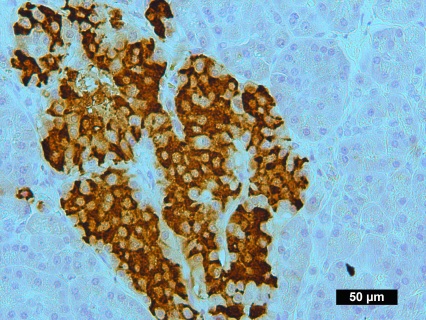
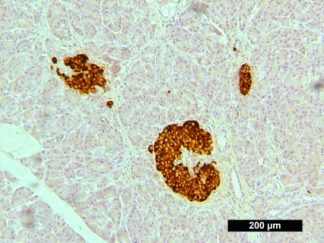
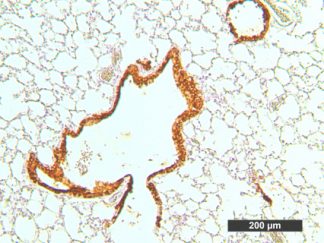
Description
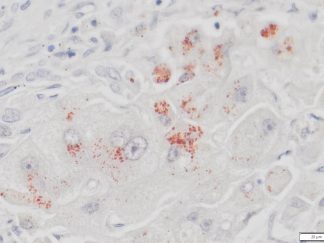
VitroView™ 1-Step Anti-Mouse Polymer-Based IHC-DAB kit is an immunohistochemistry kit for detecting a primary antibody made in mouse and visualized by 3,3′-diaminobenzidine (DAB).
Immunohistochemistry (IHC) is a method of detecting the presence of specific proteins in cells of a tissue section by exploiting the principle of antibodies binding specifically to antigens in biological tissues. IHC is widely used in the diagnosis of abnormal cells and basic research to understand the distribution and localization of biomarkers and differentially expressed proteins in different parts of a biological tissue.
Polymerizing enzymes and attaching these polymers to antibodies is a new technology. This technology has been applied to both primary antibodies and detection systems. The VitroView™ Polymer Based 1- step IHC Kit utilizes a novel polymerization technology to prepare polymeric HRP-linker antibody conjugates.
The advantages of this technology include: 1) Biotin-Free; 2) High sensitivity; 3) Low background; 4) Reduction of steps and time; 5) Ready-to-use; 6) Simplified multiple labeling.
Application
1-Step Anti-Mouse Polymer-Based IHC-DAB kit is used for immunohistochemistry for detecting a primary antibody made in mouse.
Contents
| RTU normal horse serum | 10ml |
| RTU polymeric peroxidase anti-mouse secondary antibody | 10ml |
| DAB stock solution (40×) | 0.75 ml |
| Stable DAB buffer | 30 ml |
| RTU hematoxylin solution | 10ml |
Note: RTU=ready-to-use
Reagents and Material Required but Not Provided
- Xylene and ethanol
- Distilled or deionized water
- 30% hydrogen peroxide
- 10 mM phosphate-buffered saline (PBS), pH 7.4
- Triton X-100
- Mini PAP Pen
- Primary antibody
- Mounting Media
Storage Condition
Store at 2-8°C.
Protocol
- Preparation of Slides
- For Cell Lines
- Grow cultured cells on sterile glass cover slips or slides overnight at 37 º C
- Wash briefly with PBS
- Fix as desired. Possible procedures include:
- 20 minutes with 10% formalin in PBS (keep wet)
- 10 minutes with ice cold methanol, allow to air dry
- 10 minutes with ice cold acetone, allow to air dry
- Wash in PBS
For Frozen Sections
- Snap frozen fresh tissues in liquid nitrogen or isopentane pre-cooled in liquid nitrogen, embedded in OCT compound in cryomolds. Store the frozen tissue block at -80°C until ready for sectioning.
- Transfer the frozen tissue block to a cryotome cryostat (e.g. -20°C) prior to sectioning and allow the temperature of the frozen tissue block to equilibrate to the temperature of the cryotome cryostat.
- Section the frozen tissue block into a desired thickness (typically 5-10 µm) using the cryotome.
- Place the tissue sections onto glass slides suitable for immunohistochemistry (e.g. Superfrost).
- Sections can be stored in a sealed slide box at -80°C for later use.
- Before staining, warm slides at room temperature for 30 minutes and fix in ice cold acetone or ice cold methanol for 10 minutes. Air dry for 30 minutes.
- Wash in PBS
For Paraffin Sections
- Deparaffinize sections in xylene, 3×5min.
- Hydrate with 100% ethanol, 2×2min.
- Hydrate with 95% ethanol, 2×2min.
- Rinse in distilled water.
- Follow procedure for pretreatment as required.
2. Antigen retrieval
Most formalin-fixed tissues require an antigen retrieval step before immunohistochemical staining can proceed. Heat-mediated and enzymatic antigen retrievals are common methods.
- For Citrate: Bring slides to a boil in 10 mM sodium citrate buffer, pH 6.0; maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench top for 30 minutes.
- For EDTA: Bring slides to a boil in 1 mM EDTA, pH 8.0: follow with 15 minutes at a sub-boiling temperature. No cooling is necessary.
- For TE: Bring slides to a boil in 10 mM TE/1 mM EDTA, pH 9.0: then maintain at a sub-boiling temperature for 18 minutes. Cool at room temperature for 30 minutes.
- For Pepsin: Digest for 10 minutes at 37°C.
Note: Do not use this pretreatment with frozen sections or cultured cells that are not paraffin-embedded.
- Staining Procedure
- Rinse sections in PBS-Triton X-100 (0.025%) for 2×2min.
- Serum Blocking: incubate sections with 3-4 drops of RTU normal horse serum for 30 minutes to block non-specific binding of immunoglobulin.
- Primary Antibody: incubate sections with primary antibody (mouse IgG) at appropriate dilution in antibody dilution buffer (CAT#: VB-6002) for 1-2 hour at room temperature or overnight at 4 °C. Rinse in PBS.
- Peroxidase Blocking (optional): incubate sections in 0.3% hydrogen peroxide in PBS for 10 minutes at room temperature. Rinse in PBS.
- Detection: incubate sections with 3-4 drops of RTU polymeric peroxidase anti-mouse secondary antibody for 30 minutes at room temperature.
- Rinse in PBS for 3×2min.
- Chromogen/Substrate: incubate sections with 3 drops of DAB solution for 2-8 minutes. Monitor signal development under a microscope.
Note: DAB solution is made by mixture of 25 µl of DAB stock solution with 1ml of DAB buffer.
- Rinse in distilled water 2×2 min.
- Counterstain: Incubate sections with 3 drops of RTU hematoxylin solution for 1-2 minutes. Rinse in tape water 2×2 min.
- Dehydrate through 75% ethanol for 2 min, 95% ethanol for 2 min, and 100% ethanol for 2x3min. Clear in xylene for 2×5min.
- Coverslip with mounting medium.
User Manual and Material Safety Data Sheet (MSDS) (PDF Format)