Description
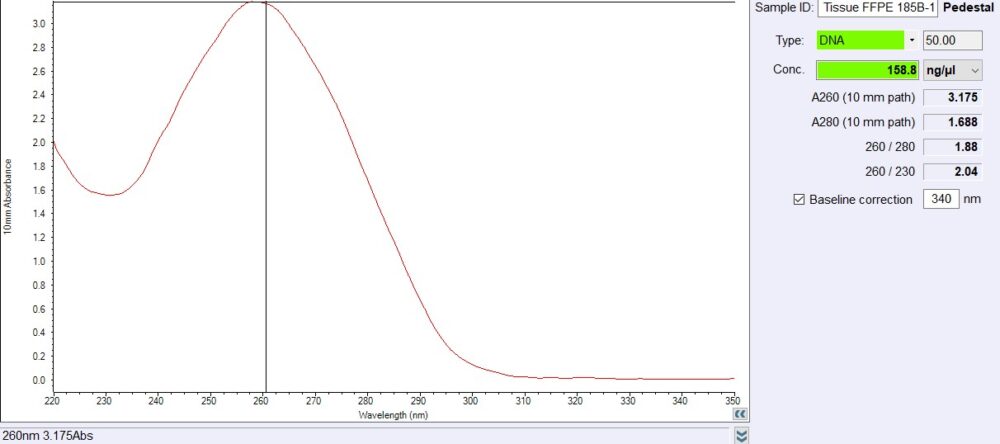
The VitroSure™ DNA FFPE Tissue Isolation Kit delivers fast, reliable, and high-yield DNA extraction from formalin-fixed, paraffin-embedded (FFPE) tissue samples. Engineered for both genomic and mitochondrial DNA isolation, this kit features proprietary VitroSure™ DNA Elute Columns and advanced silica membrane technology for consistent purification results. With flexible elution volumes (20–100 µl) and optimized performance for small tissue samples, this cost-effective, ready-to-use kit is ideal for sensitive downstream applications, including PCR, qPCR, NGS, and genotyping.
Technical Specifications
|
|
Kit Contents
|
 |
Storage
Store the Proteinase K Powder and RNase A (100 mg/ml) at -20°C. After reconstitution of Proteinase K, store the solution at -20°C. The rest can be stored at room temperature.
Procedures
- Sample preparation
1) Sample preparation from FFPE block using the Deparaffinization Solution (VB-5009, sold separately):
- Use a scalpel to trim excess paraffin off the sample block.
- Cut up to 2-6 sections with a microtome, each 5–10 µm thick. The section number for each sample depends on the tissue size. Discard the first 2–3 sections if the sample surface has been exposed to air.
- Place the sections immediately in a 1.5 or 2 ml microcentrifuge tube
- Add Deparaffinization Solution: for 2-6 sections or one 20 µm section, add 320 µl Deparaffinization Solution; for more sample material, add 640 µl Deparaffinization Solution.
- Vortex vigorously for 10 s, and centrifuge briefly to bring the sample to the bottom of the tube.
- Incubate at 56°C for 3 min, then allow to cool at room temperature (15–25°C), and centrifuge at full speed for 2 min.
- Carefully remove the supernatant by pipetting without disturbing the pellet. Carefully remove any residual Deparaffinization Solution using a fine pipette tip.
- Keep the lid open and incubate for 10 min at 37°C to dry the pellet. Proceed to step 3
2) Sample preparation from FFPE block using Xylene:
- Use a scalpel to trim excess paraffin off the sample block.
- Cut up to 2-6 sections with a microtome, each 5–10 µm thick. The section number for each sample depends on the tissue size. Discard the first 2–3 sections if the sample surface has been exposed to air.
- Place the sections immediately in a 1.5 or 2 ml microcentrifuge tube and add 1 ml of xylene to the sample. Close the lid and vortex vigorously for 10 seconds.
- Centrifuge at maximum speed for 2 minutes at room temperature.
- Carefully remove the supernatant without disturbing the pellets.
- Add 1 ml of ethanol (96–100%) to the pellet and mix by vortexing to extract residual xylene from the sample.
- Centrifuge at maximum speed for 2 minutes at room temperature.
- Carefully remove the supernatant without disturbing the pellet. Remove any remaining ethanol with a fine pipette tip.
- Open the tube and incubate at room temperature or up to 37°C for 10minutes or until all residual ethanol has evaporated. Proceed to step 3
3) Sample preparation from FFPE sections on slides:
- Submerge the slides in xylene I for 3 minutes, followed by xylene II for an additional 3minutes.
- Remove xylene by rinsing with 100% ethanol (1 minute each, repeated twice).
- Air dry the slides for 3-5minutes.
- Gently detach the tissue sections from the slides using a small blade, then transfer the tissue pellets into a 1.5 ml microcentrifuge tube.
- Proteinase K solution preparation: Combine 260 µl of Proteinase K Buffer with 5 mg of Proteinase K Powder. Vortex the mixture to ensure complete dissolution. Store the solution at -20°C.
- Resuspend the pellets in 180 µl Buffer VTL. Add 20 µl of proteinase K solution and mix by vortexing.
- Incubate at 56°C for 1 hour or until the sample is completely lysed.
- Incubate at 90°C for 1 hour without agitation.
- Cool to room temperature and briefly centrifuge the tube. For RNA-free genomic DNA, add 2 µl of RNase A (100 mg/ml) and incubate at room temperature for 2 minutes.
- Add 200 µl of Buffer VL and 200 µl ethanol to the sample. Mix thoroughly by vortexing.
- CarefullytransfertheentirelysatetoaDNAElutecolumnandcentrifugeat8000×g (or 10000 rpm) for 1 minute. Discard the flow-through.
- Add 500 µl of Buffer VDW1 and centrifuge at 8000×g (or 10000 rpm) for 1 minute. Discard the flow-through.
- Add500µlofBufferVDW2andcentrifugeat8000×g (or10000 rpm) for1 minute. Discard the flow-through and collection tube.
- Place the DNA Elute column in a clean 2 ml collection tube. Centrifuge at maximum speed for 3 minutes with the lid open to completely dry the membrane.
- 5ml microcentrifugetubeandapply20–100µl of Buffer VTE to the center of the membrane. Ensure that Buffer VTE is at room temperature.
- Incubate at room temperature for 5 minutes and centrifuge at maximum speed (20,000×g or 14,000 rpm) for 1 minute to elute the DNA.
User Manual and Material Safety Data Sheet (MSDS) (PDF)
Note: This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.
Precautions: Handle with care. Avoid contact with eyes, skin, and clothing. Do not ingest. Wear gloves.